Signaling by the nucleotide-based second messenger c-di-GMP
Despite the multitude of cues that bacteria need to monitor and respond to, the signal transduction schemes involved center on just a few types. One of the most important of these schemes is that of two component systems. A standard two component system consists of a histidine protein kinase and a partner response regulator. In response to a specific stimulus, the kinase autophosphorylates on His residue and then the phosphoryl-group is transferred to a Asp residue in the receiver domain of the response regulator.
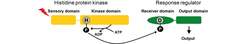
The M. xanthus genome encodes more than 200 proteins of two component systems and several of these proteins are important for fruiting body formation. Intriguingly, these genes are unusually organized, with 55% being orphan and 16% in complex gene clusters whereas only 29% display the standard paired gene organization. The large number of orphan proteins is peculiar and it represents a major challenge to understand how they are wired together. Our bioinformatics analyses have shown that a major output of signaling by two component systems is signaling by the second messenger cyclic-di-GMP.
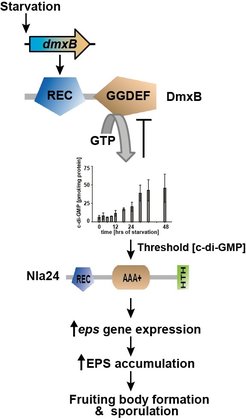
In general, c-di-GMP controls the switch from a planktonic, motile lifestyle to a surface-associated, sessile biofilm lifestyle. We have shown that c-di-GMP regulates motility in M. xanthus and that c-di-GMP is essential for fruiting body formation and sporulation. Moreover, we identified DmxB as the diguanylate cyclase (DGC) responsible for an increased c-di-GMP accumulation during development. DmxB consists of an N-terminal receiver domain and a C-terminal GGDEF domain. We found that synthesis of DmxB is regulated in response to starvation at the transcriptional level. DmxB stimulates exopolysaccharide accumulation during development and our data suggest that DmxB activity eventually results in transcription of genes responsible for exopolysaccharide synthesis. To this end, we identified the NtrC-like transcriptional regulator Nla24 as a c-di-GMP binding proteins suggesting that liganded Nla24 is involved in transcription of eps genes. DmxB can at least partially be functionally replaced by a heterologous DGC. Altogether, these results suggest a model in which a minimum threshold level of c-di-GMP is essential for successful completion of this multicellular developmental program.
Some of our recent publications on signal transduction by two component systems & the second messenger c-di-GMP:
Skotnicka, D., Smaldone, G.T., Petters, T., Trampari, E., Liang, J., Kaever, V., Malone, J.G., Singer, M. & Søgaard-Andersen, L. (2016)
A minimal threshold of c-di-GMP is essential for fruiting body formation and sporulation in Myxococcus xanthus. PLOS Genetics. 12, e1006080. doi: 10.1371/journal.pgen.1006080
Skotnicka, D., Petters, T., Heering, J., Hoppert, M., Kaever, V. & Søgaard-Andersen, L. (2016)
Cyclic di-GMP regulates type IV pili-dependent-motility in Myxococcus xanthus. J. Bacteriol. 198, 77-90. doi: 10.1128/JB.00281-15
Keilberg, D., Wuichet, K., Drescher, F. & Søgaard-Andersen, L. (2012).
A response regulator interfaces between the Frz chemosensory system and the MglA/MglB GTPase/GAP module to regulate polarity in Myxococcus xanthus. PLoS Genet. 9, e1002951. doi: 10.1371/journal.pgen.1002951
Petters, T., Zhang, X., Nesper, J., Treuner-Lange, A., Gomez-Santos, N., Hoppert, M., Jenal, U. & Søgaard-Andersen, L. (2012).
The orphan histidine protein kinase SgmT is a c-di-GMP receptor and regulates composition of the extracellular matrix together with the orphan DNA binding response regulator DigR in Myxococcus xanthus. Mol. Microbiol. 84, 147-165. doi: 10.1111/j.1365-2958.2012.08015.x
Wegener-Feldbrügge, S. & Søgaard-Andersen, L. (2009)
The atypical hybrid histidine protein kinase RodK in Myxococcus xanthus: Spatial proximity supersedes kinetic preference in phosphotransfer reactions. J. Bacteriol. 191, 1765-1776. doi: 10.1128/JB.01405-08
Shi, X., Wegener-Feldbrügge, S., Huntley, S., Hamann, N., Hedderich, R. & Søgaard-Andersen, L. (2008)
Bioinformatics and experimental analysis of proteins of two-component systems in Myxococcus xanthus. J. Bacteriol. 190, 613-624. doi: 10.1128/JB.01502-07

