Collective Motion of Escherichia coli at High Cell Density
Density effects impair chemotactic behavior
When the cell density of a suspension of swimming bacteria increases, collective motion emerges, characterized by intermittent jets and swirls of groups of cells. Our analysis of bacterial swimming in controlled gradients of a chemical attractant shows that chemotaxis is strongly reduced as collective motion develops, suggesting that collective reorientations interfere with the chemo-sensing mechanism.

Collective motion in bacterial suspensions. At the right: Typical snapshot of the velocity field measured in a high-density cell suspension.
Introduction
In dilute suspension, bacteria swim in relatively straight second-long runs interrupted by short reorientations (tumbles), resulting at long times in a random walk by which they explore their environment. Bacteria monitor—via their chemoreceptors—the changes in environmental conditions and respond to them by modifying a phosphorylation signal transmitted to the flagellar motors to change the tumbling frequency accordingly. The biochemical signalling pathway controlling this chemotactic behaviour is well understood in E. coli and it is one of the best modelled biological signalling systems. In E. coli, attractant substances repress the phosphorylation signal, which results in prolonged runs, repellents having the opposite effect. An adaptation module slowly resets the receptor sensitivity for further stimulations, via a negative feedback loop. This effectively allows the cell to compare its current situation to the recent past while swimming along a given direction, with a memory time scale of a few seconds.
In high-density populations, collective motility takes over
When the cell density of a suspension of swimming bacteria increases, collective motion emerges, characterized by intermittent jets and swirls of groups of cells. This behaviour is observed for many microorganisms not only in artificial but also in natural situations, often at an interface, e.g., when bacteria swarm on a moist surface in the lab or during infection, or at an air-water interface during formation of pellicle biofilms.
Bacterial collective motion has been extensively studied experimentally and theoretically, and it is known to emerge from the alignment between the self-propelled cells. Two alignment mechanisms have been proposed, based either on steric interactions between the rod-like bacteria or on the hydrodynamics of the flow they create as they swim. However, the relative importance of these two mechanisms has not been clearly established so far.
Collective reorientations impair chemotaxis
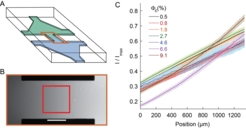
Microfluidic device used for chemotaxis assays. A) Schematic representation of the device, where two reservoirs with different chemical composition are connected by a channel in which the chemoattractant gradient forms. B) Central part of the device, highlighted orange in (A), with gradient profile across the channel quantified using fluorescein. Scale bar is 500 μm. The red box indicates the location at which cellular behaviour is recorded. C) Examples of gradients, measured in the h=50 μm device, for cell suspensions of indicated cell densities. Error bars represent measurement error.
To measure both the collective dynamics and the chemotactic response of populations of E. coli cells at varying density, we analyzed bacterial swimming in controlled gradients of a chemical attractant using a microfluidics setup, video microscopy and Fourier image analysis.
We observed that the size of bacterial flow structures in fully developed collective motion is set by the smallest system size, the channel height, independently of cell length and volume fraction, and that an increasing amount of the kinetic energy of the system gets poured into this flow structure as the cell density increases. This property strongly suggests that hydrodynamics plays the primary role in the emergence of the collective motion.
The experimental results showed that, after increasing up to a maximum at intermediate densities, chemotaxis is strongly reduced as collective motion develops, suggesting that collective reorientations interfere with the chemo-sensing mechanism. Physical interactions between cells at high densities result in a strong reduction and ultimately in the abolishment of the specific ability of E. coli to track chemical gradients. Collective motion acts directly on the mechanism of gradient-sensing since the gradient itself is little affected. Our analysis based on agent-based simulations suggests that this reduction is induced by an increased reorientation rate of cell bodies due to the collective motion.
Density effects have wide consequences for bacterial populations in the wild
These results have important implications for collective behaviours of motile bacteria. First, the observed regulation of chemotactic behaviour through physical interactions among motile cells it provides a physical mechanism that might regulate chemotactic accumulation of bacteria near sources of chemoattractants (e.g., nutrients), because gradually increasing cell density would initially promote and subsequently limit the process. This cap on the number of cells which can reach a given food source or cell patch should play an important role for resource distribution within bacterial populations, as well as their self-organization.
Second, the observed strong reduction in chemotactic drift at cell densities typical of swarming suggests that, without specific counteracting mechanisms, chemotactic navigation of bacteria swimming within a swarm is nearly impossible. Although more prominent steric interactions within a swarming colony might potentially improve tracking of gradients at high density, as could other differences in swimming behaviour, or additional cohesive interactions caused by cell differentiation in a swarm, our results suggest that the emergence of swirling collective motion fundamentally undermines the chemotactic behaviour.














