And … Action!
Atomic Crystal Structure of Activated [Fe]-hydrogenase Resolved
The limitation of conventional energy resources poses a major societal challenge and requires the development of environmentally friendly engines or fuel cells. In this context molecular hydrogen (H2) is regarded an important energy carrier for future applications. Nature uses hydrogenase enzymes to mediate production and consumption of H2. Presumably having evolved in bacteria long before the earth's atmosphere contained O2, most of these bacterial hydrogenases are very sensitive to O2, which obviously makes them difficult to handle for technology. Researchers in the field of synthetic biochemistry aim to design new, robust synthetic enzyme models for technical purposes. For this, the exact knowledge of structure and function of the catalytic center of these enzymes is of fundamental importance.
The [Fe]-hydrogenase catalyzes H2-activation and hydrogenation reactions using a unique prosthetic group, the FeGP cofactor. A decade ago, the Shima group has reported the crystal structure of this enzyme in an open inactive form (Shima et al. Science, 2008, 321: 572). Based on this work, the researchers were able to synthesize many mimic models of the cofactor chemically. Thus, the FeGP cofactor proved to be an excellent template for designing new hydrogenation and dehydrogenation catalysts. However, in order to construct highly efficient catalysts, more detailed information on the activated [Fe]-hydrogenase was necessary.
![Based on the X-ray structure of [Fe]-hydrogenase in an active conformation, computational simulations provide precise insights into H2-activation. This knowledge is important for exploiting [Fe]-hydrogenase for technological purposes such as hydrogenation reactions, production of H2 or using H2 as a fuel.](/619688/original-1561360749.jpg?t=eyJ3aWR0aCI6MjQ2LCJvYmpfaWQiOjYxOTY4OH0%3D--fc17e8b6fae183cbdd1818c280d2cdaf5bd83168)
Based on the X-ray structure of [Fe]-hydrogenase in an active conformation, computational simulations provide precise insights into H2-activation. This knowledge is important for exploiting [Fe]-hydrogenase for technological purposes such as hydrogenation reactions, production of H2 or using H2 as a fuel.
Seigo Shima and his group pursued this task in collaboration with scientists from the Max Plank Institute of Biophysics in Frankfurt, Max Planck Institute for Chemical Energy conversion, Freie Universität Berlin and École Polytechnique Fédérale de Lausanne. After 10 years of research, the Max-Planck researchers have succeeded in obtaining the atomic-resolution (1.06 Å) crystal structure of the [Fe]-hydrogenase in the substrate-bound state. Upon substrate (methenyl-H4MPT+) binding, the active-site cleft closes and the water ligand of the Fe site is kicked out from the Fe center. The removal of the water ligand creates an open Fe site for the H2 binding, which is ready for H2-cleavage. “Based on structural analyses, we illustrated the catalytic cycle of the [Fe]-hydrogenase”, Seigo Shima explains. “Our finding provides chemists with indispensable information for designing new robust H2-activation/generation catalysts.”
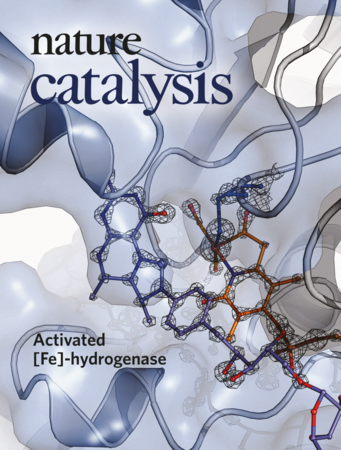
![Based on the X-ray structure of [Fe]-hydrogenase in an active conformation, computational simulations provide precise insights into H2-activation. This knowledge is important for exploiting [Fe]-hydrogenase for technological purposes such as hydrogenation reactions, production of H2 or using H2 as a fuel. Based on the X-ray structure of [Fe]-hydrogenase in an active conformation, computational simulations provide precise insights into H2-activation. This knowledge is important for exploiting [Fe]-hydrogenase for technological purposes such as hydrogenation reactions, production of H2 or using H2 as a fuel.](/619688/original-1561360749.jpg?t=eyJ3aWR0aCI6ODQ4LCJmaWxlX2V4dGVuc2lvbiI6ImpwZyIsIm9ial9pZCI6NjE5Njg4fQ%3D%3D--35286a94fd452601855374634788f26e7dfecdc5)











