Smut disease
Smut diseases are caused by a large group of biotrophic fungal pathogens which parasitize mostly grasses including a number of cereal hosts like maize, barley and wheat. They are characterized by a narrow host range, infect plants systemically and usually cause symptoms in male and female inflorescences only. The fungus Ustilago maydis causing smut disease in maize is an exception, however. U. maydis symptoms are large tumors that can develop on all above ground parts of the plant. In the field the most prominent tumors develop in the cobs (Figure 1A). As plant nutrients are reallocated to producing fungal biomass, infected plants show stunted growth and a significant reduction in crop yield. The ability of U. maydis to cause symptoms on all aerial organs of the plant, its highly efficient homologous recombination system that can be used to generate deletion mutants, its small genome and the many versatile molecular tools developed, have made the U. maydis-maize interaction one of the most attractive models for studying biotrophic plant pathogenic fungi and for elucidating the mechanisms underlying disease

Figure 1A: From the field to the laboratory. A) U. maydis infected maize cobs were collected in a field near Amöneburg in September 2015. B) In the laboratory tumors that have developed in kernels are removed for a subsequent biochemical analysis.
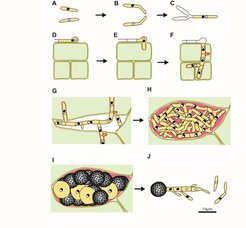
In all smut fungi pathogenic development is initiated by the filamentous dikaryon that forms infection structures (appressoria) on the leaf surface, allowing direct penetration of the cuticle. During penetration the plasma membrane of the host invaginates and encases the infection hyphae, shielding the fungus from direct contact with the host cytoplasm and creating an extended interaction zone. Although biotrophic U. maydis hyphae traverse plant cells, they elicit only limited host defense responses and the infected plant tissue remains alive until late in the infection process. At that stage fungal proliferation occurs mostly in the apoplast and leads to huge fungal aggregates which are embedded in a polysaccharide matrix. In these aggregates hyphae fragment and differentiate into the diploid spores (Figure 1B).
Initial plant defense responses triggered by fungal PAMPs are actively suppressed by U. maydis during colonization. This is accomplished by a battery of several hundred secreted effector proteins that either have a function in the tight interaction zone between fungal hyphae and host plasma membrane (apoplastic effectors) or are translocated into host cells and modulate endogenous plant processes (cytoplasmic effectors). The majority of the U. maydis effector proteins are novel, i.e. they lack characteristic motifs that could give hints to their function. Effector gene expression is usually tightly coupled to the biotrophic phase. So far, the molecular function of only six U. maydis effectors has been elucidated.