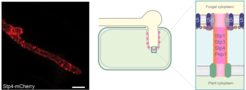
A fungal surface-exposed protein complex likely involved in effector delivery to the host
By co-IP of three effectors (Stp1, Stp2 and Stp3) essential for virulence from infected plant material, we found these three effectors in a complex with two additional effectors (Stp4 and Pep1) and two fungal transmembrane proteins (Stp5 and Stp6). This seven-protein-complex termed Stp complex resides in the plasma membrane of U. maydis and extends into host cells where it contacts proteins in the host plasma membrane that tightly encase biotrophic fungal hyphae. Every single component of the complex is critical for plant colonization and mutants become arrested in the epidermal layer of maize. Mutants lacking the complex fail to downregulate plant defense responses including plant cell death and fail to induce nonhost resistance. All these processes depend on the delivery of fungal effectors to the host and therefore we implicate the Stp complex in effector translocation (Ludwig et al., 2021).