Protein diffusion barriers
Eukaryotic cells are characterized by an abundance of subcellular compartments that are separated from the rest of the cell by membranes or proteinaceous diffusion barriers. Whereas specialized membrane-bounded compartments are also found in some bacteria, other compartmentalization mechanisms were thought to be absent from prokaryotic cells. However, studying protein diffusion in C. crescentus cells, we have recently identified a multi-protein complex that serves as a general protein diffusion barrier, preventing the exchange of proteins between the stalk and the cell body (Schlimpert et al., 2012).
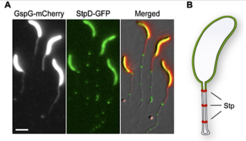
The C. crescentus stalk is an extension of the cell body that largely consists of cell envelope surrounding a thin thread of cytoplasm and grows by insertion of new cell wall material at the stalk base. It is interrupted at regular intervals by so-called crossbands, disc-shaped electron-dense structures whose nature and function had remained unknown. We found that these crossbands are polymeric complexes composed of at least four different proteins (StpABCD), which extend from the cytoplasmic core of the stalk to its outer membrane layer. Blocking diffusion of both membrane and soluble proteins, they physiologically separate the stalk from other parts of the cell and thus ensure that newly synthesized proteins are retained in the cell body. As a consequence, cells are able to adapt faster to environmental changes that require the de novo synthesis of proteins. Moreover, crossbands enable cells to save energy by reducing the amount of protein required to achieve optimal working levels, thereby providing a fitness advantage in the oligotrophic environment that C. crescentus inhabits. In the future, this machinery could be adapted for generating artificial cellular compartments that may facilitate the storage of toxic biosynthetic products.
